How do organisms keep track of time and what determines the lifespan of a species?
The mechanisms that underlie biological timing remain largely unknown. Despite the high conservation of genetic programs throughout the animal kingdom, the duration of embryogenesis and lifespan are species-specific. For instance, mouse development lasts around 20 days, and the embryonic period of human gestation takes place during the first 60 days of pregnancy. These differences in timing arise at conception, as the progression from the fertilized zygote to embryo implantation lasts around four days in mouse whereas it takes seven days in human. Further, during diapause some species can halt development for extended periods of time with no apparent trade-offs for development or lifespan.
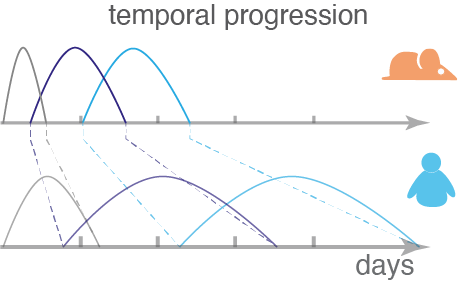
Our lab studies the regulatory and dynamic processes that control timing in development and homeostasis across and within species with the long-term goal to modulate biological timing in a precise and tunable manner. Our current research questions are:
- What controls biological timing?
- Can we modulate developmental timing and extend lifespan?
- What is the role of protein turnover in developmental timing and lifespan?
We make use of comparative human and mouse stem cell models as well as embryos to search for the regulatory mechanisms that determine species-specific timing. The lab employs genetic and pharmacological manipulations and quantitative and temporally resolved techniques such as flow cytometry, imaging, and genome-wide approaches to investigate the molecular and metabolic mechanisms that regulate developmental timing.
Overall, the identification of physiological mechanisms that modulate timing and its translation to stem cell models may have important implications in the field of human assisted reproduction, regenerative medicine, and aging. Changing the pace of developmental processes may facilitate the generation of clinically relevant cell types faster or it may allow lifespan extension.
